Electrical Conductivity Of Water Pdf

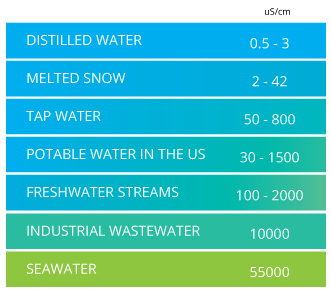
Conductivity of water water type conductivity umhos cm distilled water 0 5 3 0 melted snow 2 42 potable water in u s.
Electrical conductivity of water pdf. Distilled water has greater conductivitythan pure water because it is always contaminated with atmospheric carbon dioxide which dissolves in water to form the weak electrolyte carbonic acid. Electrical conductivity s is the reciprocal of electrical resistance ω. Electrical conductivity is measured in microsiemens per centimeter us cm. In general the measurement of conductivity is a rapid and inexpensive way of determining the ionic strength of a solution.
While the electrical conductivity is a good indicator of the total salinity it still does not provide any information about the ion composition in the water. Many ec meters nowadays automatically standardize the readings to 25 o c. Water quality standards electrical conductivity is a measure of the saltiness of the water and is measured on a scale from 0 to 50 000 us cm. 2015 this is also observed as the ec showed strong.
High purity water resistivity conductivity measurement ultra pure water without any chemical impurities will still have a conductivity because of the presence of h and oh ions due to the self ionization of water. Conductivity can be much higher than the maximum values shown above under. Conductivity measurements cover a wide range of solution conductivity from pure water at less than 1x10 7 s cm to values of greater than 1 s cm for concentrated solutions. S ω 1 electrical conductivity is normally expressed in units of ms m 1.
Electrical conductivity shows the current conductance ability of water and also has direct relationship with tds in water mihir et al. Water the greater its electrical conductivity. When you place a conductivity sensor in the water a mild electrical current is passed between two electrodes. The electrical conductivity of water increases by 2 3 for an increase of 1 degree celsius of water temperature.
Total dissolved solids tds in mg l 0 64 x electrical conductivity in µmho cm. In the two electrode conductivity mea. Assuming the conductivity of a solution is mainly due to the dissolved solids the value of electrical conductivity can be used to get an indirect result of total dissolved solids by the following equation. Ultra pure water has a conductivity of 0 055 µsiemens cm or a resistivity of 18 18 mω cm at 25 c.
30 1500 freshwater streams 100 2000 the table above shows some ranges of conductivity values you might encounter in the field. The graph shows the conductivity of pure water distilled water and typical electrolytes at 25 c. Conductivity can be regarded as a crude indicator of water quality for many purposes since it is related to the sum of all ionised solutes or. If you measure an increase in µs cm you are measuring an increase in conductivity of the water.